|
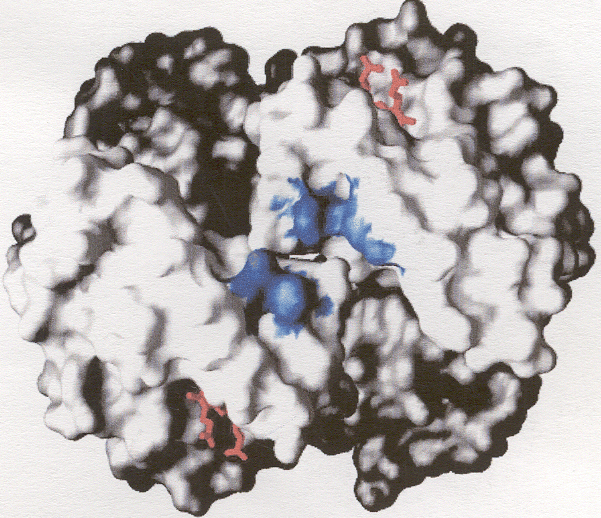
Proteins localized in thin clay films alter both the thermodynamics and kinetics of electron transfer reactions by the protein with the underlying electrode. We hypothesize that this is related to stabilization of conformations of the bulk globin by electrostatic interactions with the clay surfaces. The Figure at the left shows a GRASP computation of Myoglobin with electrostatic positive charges in blue and negative charges in red. The cleft showing the opening to the heme site indicates that this side of the protein is predominately postively charged at neutral pH values. The red sites are located at the "edges" of the protein and may help orient the globin with respect to the clay surface, and, therefore with respect to the underlying electrode surface.
This work derives from earlier work with the kinetics of electron transfer events at cross-linked hemoglobins demonstrating that changes in the globin structure affect both the potential (thermodynamics) of the reaction and the rate of the reaction by altering access of water to the heme site in the hemoglobin.
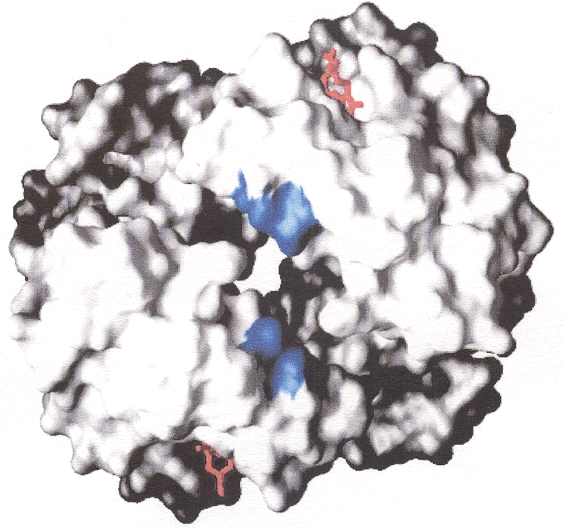
The image at the left represents the R and T forms of hemoglobin with the positive change shown, which alters intensity and location with conformational change imposed by crosslinking.
|